Microbial influenced corrosion (MIC)
 Although corrosion is an electrochemical process, it is caused or accelerated in >50 % of the cases by microbiological activity. Microbial corrosion can be prevented, but is a complex problem once it is there. In short it comes down to the following: The corrosion process often takes place under a protective biofilm that can be thin, thick, hard, slimy or tough. Under the biofilm, micro-organisms often live in symbiosis with each other, aerobic and anaerobic.
Although corrosion is an electrochemical process, it is caused or accelerated in >50 % of the cases by microbiological activity. Microbial corrosion can be prevented, but is a complex problem once it is there. In short it comes down to the following: The corrosion process often takes place under a protective biofilm that can be thin, thick, hard, slimy or tough. Under the biofilm, micro-organisms often live in symbiosis with each other, aerobic and anaerobic. 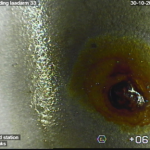 One often cares for the nutrients of the other. If the development goes fast enough (among other things under the influence of the right temperature, sufficient nutrients) the pH decreases dramatically, causing the metal to go into solution very locally. In a system with stagnant water (for example sprinkler system or water that remains behind in pipes and tanks after a hydrotest) leakage can occur within a few months. MIC can develop 10 – 100 times faster than a normal electro-chemical proces. Free bacteria are easy to fight, but the microorganisms under the biofilm are not. The focus must be on complete removal of the biofilm.
One often cares for the nutrients of the other. If the development goes fast enough (among other things under the influence of the right temperature, sufficient nutrients) the pH decreases dramatically, causing the metal to go into solution very locally. In a system with stagnant water (for example sprinkler system or water that remains behind in pipes and tanks after a hydrotest) leakage can occur within a few months. MIC can develop 10 – 100 times faster than a normal electro-chemical proces. Free bacteria are easy to fight, but the microorganisms under the biofilm are not. The focus must be on complete removal of the biofilm.
One metal is more sensitive than the other, but nothing is really resistant to MIC: stainless steel; carbon steel; cemented steel; galvanized, aluminum, cunifer … even copper. Also in piping systems with sufficient flow > 2 mtr / sec) there may be dead spots where MIC can easily develop. In the winter also in inactive heat exchangers, in / on the heatings coils at the bottom of a storage tank, or on underwater constructions such as jetties and bollards.  The source may as well be river water as potable water in which sulphate reducing bacteria. Harmless to humans, but potentially disastrous for a stainless steel production line.
The source may as well be river water as potable water in which sulphate reducing bacteria. Harmless to humans, but potentially disastrous for a stainless steel production line.
If MIC is suspected, it requires a thorough and professional investigation because ….
The presence of micro-organisms involved in corrosion does not imply that there is also MIC!
MIC is not easy to get rid of and requires a careful, decisive approach. This can be costly and sometimes very awkward from an operational point of view. That is why you want to be sure that treatment is really necessary. The ‘MIC conclusion’ is sometimes too lightly drawn. The treatment is then unnecessarily expensive or not at all effective because not MIC but another corrosion mechanism is the issue. As with so many other corrosion mechanisms, it is the interpretation of the investigation that determines which measures are adequate. The Corrosion Control Technology Centre consider microbial influenced corrosion to one of its specialties. Not because all knowledge is present at one person. But because different disciplines are taking care of the phenomenon. MIC is only MIC when our microbiologist, our chemical technologist and our metallurgist agree. The corrosion alliance needs independent proof before a solution is provided. In other words MIC is only proven when not only the bacteria in question have been demonstrated, but also that their involvement in the corrosion problem has been proven both chemically and metallurgically. Moreover, the development potential must be examined on the basis of a nutrient analysis and not least, what the cause is or the source. And once MIC has been proven, what form of treatment is possible and prevention? Because before you know it, the problem is back. Sometimes micro-organisms can not be eradicated and the equipment must be protected. Other specialist disciplines are also needed for this. MIC stirs up many questions and as many answers. You can find them at the Corrosion Control Technology Center.
Examples of bacteria that can be involved in the corrosion proces:
- Sulphate reducing bacteria
- Sulphur oxidising bacteria
- Iron reducing bacteria
- Iron oxidising bacteria
- Methane producing micro-organisms
When it comes to ‘biocorrosion’ prevention is much better than cure. Because once biocorrosion is present, it is very difficult to cure it for 100%. Thanks to the development of a practical method whereby the risks of biocorrosion are monitored, the risks of damage can be controlled. With preventive measures, timely action can be taken at the moment that the threat is relevant.
The Corrosion Control Technlogy Alliance, in collaboration with renowned partners, offers a comprehensive program to prevent and cure microbial corrosion. Download the brochure

Corrosion Control Technology Centre
Contact: Frans van der Kolk
T. +31 (0)653206225
kolk@corrosioncontrol.nl
